- info@buhlmannlabs.ch
- +41 61 487 12 12
- Home
- Products & Solutions
- Gastroenterology
- Calprotectin

CALPROTECTIN - The ideal tool to diagnose and monitor IBD patients
Calprotectin is the best marker for IBD. It is an established measure to discriminate IBD from IBS and a very valuable tool to monitor the disease course of IBD patients (Crohn’s and ulcerative colitis). BÜHLMANN Laboratories offers the largest product portfolio in the market of calprotectin testing and has set the standard for more than 10 years.

CALPROTECTIN - The ideal tool to diagnose and monitor IBD patients
Calprotectin is the best marker for IBD. It is an established measure to discriminate IBD from IBS and a very valuable tool to monitor the disease course of IBD patients (Crohn’s and ulcerative colitis). BÜHLMANN Laboratories offers the largest product portfolio in the market of calprotectin testing and has set the standard for more than 10 years.
The BÜHLMANN fecal Calprotectin Assays – A unique Success Story
Calprotectin – Differentiating IBD from non-inflammatory Diseases
Inflammatory Bowel Disease (IBD) affects approximately 2 million people in Europe and shows increasing prevalence all over the world. Crohn’s Disease (CD) and Ulcerative Colitis (UC) are incurable serious chronic diseases of the intestinal tract. Symptoms of IBD strongly resemble those of non-inflammatory diseases such as the irritable bowel syndrome (IBS), polyps or even gastrointestinal infections. Calprotectin has proven itself as a very good surrogate marker in the diagnosis of IBD and helps therefore to reliably select patients for further invasive diagnostic procedures.
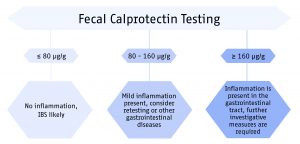 Suggested algorithm for the diagnostic procedure using calprotectin as surrogate marker for intestinal inflammation in IBD. Calprotectin values below 80 µg/g are not indicative for an active inflammation in the gastrointestinal tract. Calprotectin values of 80 – 160 µg/g can represent mild organic disease such as inflammation caused by NSAIDs, mild diverticulitis and IBD in remission phase. Values above 160 µg/g are highly indicative for active inflammation in the gut.
Suggested algorithm for the diagnostic procedure using calprotectin as surrogate marker for intestinal inflammation in IBD. Calprotectin values below 80 µg/g are not indicative for an active inflammation in the gastrointestinal tract. Calprotectin values of 80 – 160 µg/g can represent mild organic disease such as inflammation caused by NSAIDs, mild diverticulitis and IBD in remission phase. Values above 160 µg/g are highly indicative for active inflammation in the gut.
Calprotectin – Monitoring IBD Disease Activity
Calprotectin Product Information
| Name | BÜHLMANN fCAL® ELISA | BÜHLMANN fCAL® turbo | Quantum Blue® fCAL |
| Method | ELISA | Immuno turbidimetric | Lateral Flow |
| Time to Result | 2 h (approx.) | 10 min (approx.) | 15 min (approx.) |
| Sample Type | 50-150 µl feces extract | less than 1 g | 60 µl feces extract |
| Standard Range | 10-600 µg/g / 30-1800 µg/g | 0-2000 µg/g | 100-1800 µg/g / 30 -1000 µg/g |
| Sensitivity (LoQ) | 10 µg/g | 22 µg/g | <100 µg / <30 µg/g |
| Order Code | EK-CAL 96 wells | B-KCAL-RSET, B-KCAL-CASET, B-KCAL-CONSET | LF-CHR25 / LF-CALE25 (25 tests each) |
| CE (IVDR compliant) | CE (IVDR compliant) | CE/CE (IVDR compliant) |
Calprotectin – Stool Extraction with CALEX®
BÜHLMANN Laboratories is not only leader in unique diagnostic assays for calprotectin, but has also the most innovative stool preparation device demonstrating the importance of pre-analytics in calprotectin measurement. The CALEX® offers simple and hygienic stool sampling and is pre-filled with the BÜHLMANN extraction buffer. Check out our main page for stool preparation now

Social Links