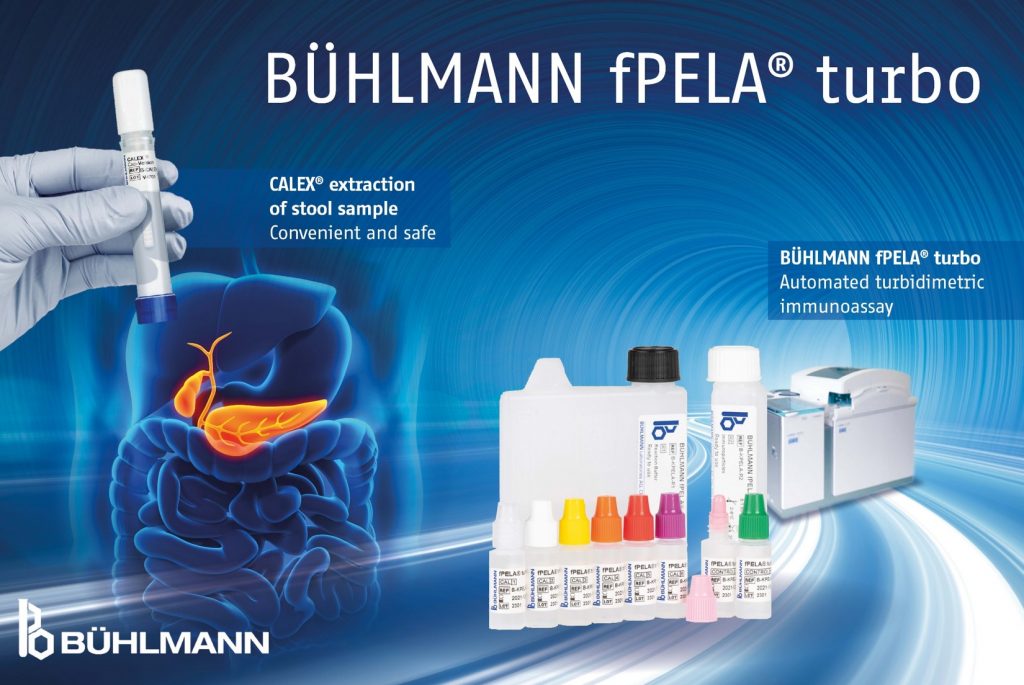
BÜHLMANN proudly announces the availability of the new turbidimetric assay for the simplified quantification of human pancreatic elastase, the BÜHLMANN fPELA® turbo.
The determination of pancreatic Elastase levels is the most commonly employed indirect test for exocrine pancreatic function. Pancreatic exocrine insufficiency (PEI), is a condition in which patients are unable to adequately digest fats, carbohydrates and proteins due to a lack of digestive enzymes secreted by the pancreas. This results in nutrient malabsorption and malnutrition with severe consequences in the quality of life.
- The turbidimetric assay developed by BÜHLMANN facilitates the automation of the well-established but work intensive manual ELISA method.
- The turbidimetric assay is developed to be run directly with stool extracts derived from CALEX® Cap extraction device. The same buffer as for fecal calprotectin is applied. Therefore, both assays can be run from the same single extraction.
- So far applications have been established already on the cobas c501/c502 from Roche, the Alinity and Architect analyzer from Abbott as well as on the AU analyzers from Beckmann. Further applications are in planning and will come soon.
Market release of the certified CE IVD product is planned in Spring 2020.

Social Links